A recent report published by the International Energy Agency states that meeting the Paris agreement’s climate targets would send demand skyrocketing for the “critical minerals” used to produce clean energy technologies. The figures are particularly dramatic for the raw materials used to manufacture electric vehicles: by 2040, the IEA forecasts that demand for lithium will have increased 42 times relative to 2020 levels.
Lithium batteries are commonly used for portable electronics and electric vehicles and are growing in popularity for military and aerospace. Some would say ‘The lithium-ion battery is an epoch-making invention’.
However, lithium batteries also contain a flammable electrolyte.
The Samsung Note 7, the device banned from flight by the FAA, is “only a symptom of a problem with all lithium ion batteries,” Cox told the standing-room-only crowd. “We’re flying more and seeing more devices on airplanes. It’s going to come up again.”
Effective April 1, 2016, more stringent regulations were issued by the International Air Transport Association (IATA) for the transport of Lithium Batteries that are packed and shipped as loose/bulk (UN3480/PI965).
Types of lithium batteries
There are many different types of lithium batteries. The three main types are described here.
Lithium-ion batteries
Lithium-ion batteries (Li-ion or LIB batteries) have lithium compounds as the electrode material, and are rechargeable. Li-ion batteries are widely used in portable electronic products such as mobile phones, laptops, tablets, MP3 players and cameras.
Lithium-metal batteries
Lithium metal batteries have lithium metal as an anode and are generally not rechargeable. They come in different shapes and forms, including the flat, round batteries used in watches. They are also commonly used in products such as calculators or torches.
Lithium-ion polymer batteries
Lithium-ion polymer batteries, often called lithium polymer batteries (Li-poly, Li-Pol, LIP, PLI or LiP), are rechargeable batteries usually composed of several identical secondary cells in parallel.
They are used in some portable electronic products and fall under the family of lithium-ion batteries.
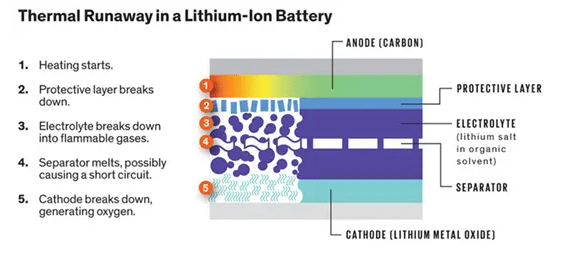
There’s no way to predict when a thermal runaway is going to occur, igniting a lithium battery fire. A thermal runaway starts from an internal short that may be caused by a manufacturing defect, physical damage or heat.

A lithium battery fire also releases an ether-based vapor that’s highly flammable. The chemical reaction of a thermal runaway can release hydrogen and oxygen byproducts, “So this process creates its own fuel, its own ignition and its own oxygen,” said Cox.

Thermal runaway can occur due to an internal short circuit caused by physical damage to the battery or poor battery maintenance. The same type of scenario could cause an external short circuit which could also kick off the chain reaction.
Battery users not only need to handle and use their batteries carefully, but they need to replace them as well. This is because the chemicals and materials degrade over time.
If you have an old battery that has been uncharged or undercharged, it may have built up gasses within the casing. This state can easily cause a battery to explode.
If you see a deformed or “bubbled” battery, do not attempt to charge it. Properly dispose of and replace any deformed batteries.

In Australia it was reported that one of the giga batteries was on fire, giving off toxic fumes. It stated:
A Tesla battery has burst into flames during testing at the site of the southern hemisphere’s largest battery project.
A 13-metric-ton lithium battery caught fire on Friday at the renewable energy plant, called the Victorian Big Battery, near Geelong, about 50 miles from Melbourne. The blaze then spread to an adjacent battery bank, Australia’s ABC reports, but has since been contained.
A toxic smoke warning has been issued in the area. Fire crews will have to wait up to 24 hours for the blaze to die down.
The site is the second Tesla battery project Down Under, following the 2017 installation in South Australia, a facility which Tesla CEO Elon Musk called the “world’s largest” at the time.
Previous blogs, such as the one below, show the process of acquiring non-ferrous metals to create lithium batteries.

Pingback: The Sand is Running Out | borderslynn
Pingback: Lithium – the new gold vs. historic spiritual land | borderslynn